Standard operating Procedure for Operation, Calibration, and cleaning of the Tablet Hardness tester in pharmaceuticals.

1.0 OBJECTIVE: To establish the procedure for the operation, calibration, and cleaning of the tablet hardness tester.
2.0 SCOPE: This SOP applies to the tablet hardness testers within the Quality Control department.
3.0 RESPONSIBILITY: Officer, Executive – Production Department
Manager – Production Department
QA Officer – In process
4.0 DEFINITIONS:
NA (Not Applicable)
5.0 PROCEDURE:
5.1 Operation:
5.1.1. Verify and ensure that the hardness tester is calibrated.
5.1.2. Position the tablet between the fixed and moving jaws in an edgewise orientation.
5.1.4. Slowly turn the screw knob clockwise until the tablet fractures.
5.1.5. Record the measurement displayed on the scale.
5.1.6. Document the hardness result in kilograms per square centimeter.
5.1.7. Release the screw knob by turning it anti-clockwise.
5.1.8. Confirm that the holding jaw is clear of any tablet residue before placing a new tablet for testing.
5.2 Calibration:
5.2.1. Ensure that the tablet hardness tester is clean.
5.2.2. Attach the device for affixing standard weights to the hardness tester’s pointer.
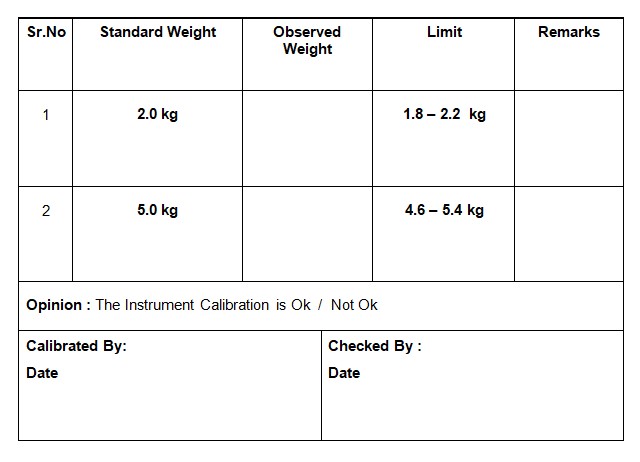
5.2.3. Affix the standard weights to the attachment and record the scale reading in Annexure-I.
5.2.9. If the readings exceed acceptable limits, tag it as “OUT OF CALIBRATION” and contact the service engineer.
5.2.10. Utilize standard weights certified by the Weights and Measures Department.
5.2.11. Calibration is conducted once every six months.
Related SOP: SOP on Metal Detector and Vertical Deduster
5.3 Cleaning:
5.3.1. Clean the hardness tester by wiping it with a clean, dry cloth. Follow this by wiping with a cloth dampened with potable water, and finish by drying it with a clean, dry cloth.
6.0 ABBREVIATIONS:
Nil
7.0 REFERENCES:
NA
8.0 ANNEXURE:
Annexure-I – Calibration Record: Tablet Hardness Tester